 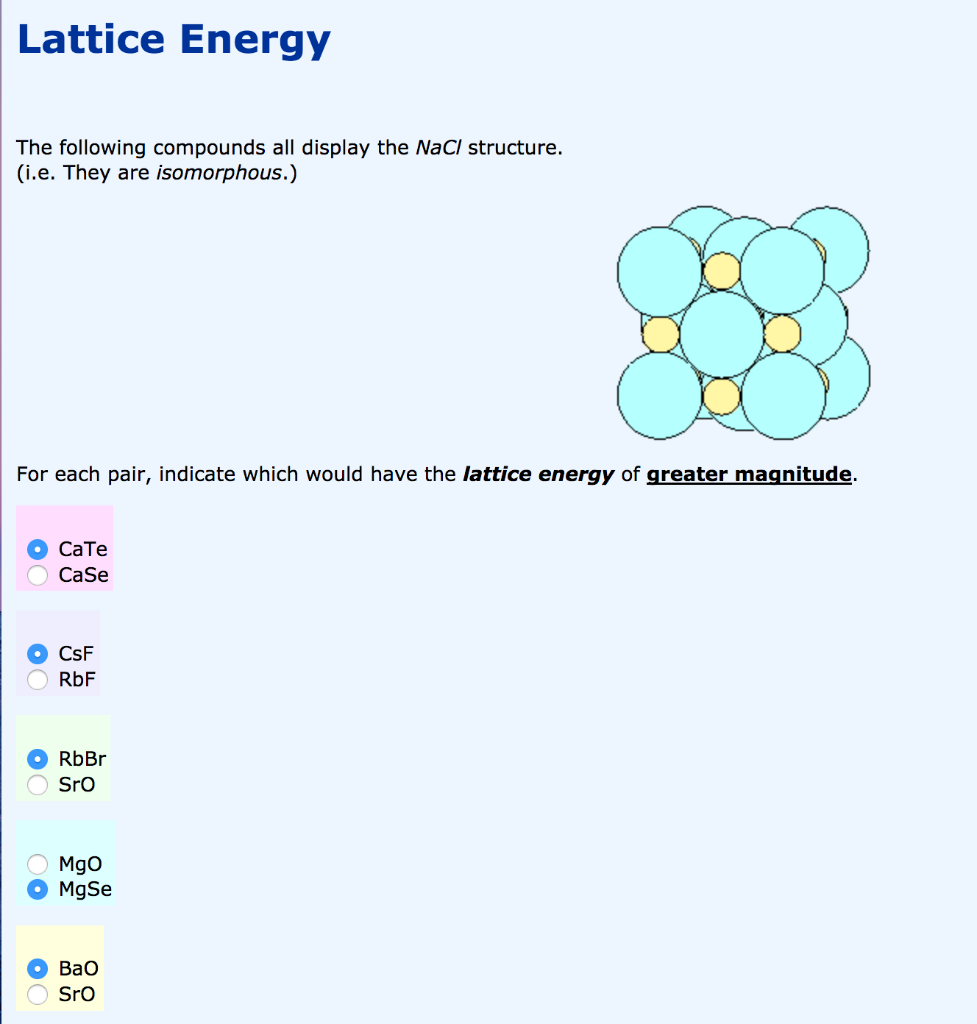
These salts all actually exhibit the sodium chloride lattice (CN 6). ΔH 3 = dissociation energy of Cl 2(S) = 244 kJ mol –1 1 atom at the corners, a total of 4 atoms. ΔH 2 = ionisation energy of Na(S) = 495.0 kJ mol –1 ΔH 1 = heat of sublimation of Na(S) = 108.7 kJ mol –1 ΔH f = heat of formation of sodium chloride = – 411.3 kJ mol –1 Let us calculate the lattice energy of sodium chloride using Born-Haber cycle The sum of the enthalpy changes of these steps is equal to the enthalpy change for the overall reaction from which the lattice enthalpy of NaCl is calculated. Also, the formation of NaCl can be considered in 5 steps. Since the reaction is carried out with reactants in elemental forms and products in their standard states, at 1 bar, the overall enthalpy change of the reaction is also the enthalpy of formation for NaCl. Let us use the Born - Haber cycle for determining the lattice enthalpy of NaCl as follows: ˆ†H f - enthalpy change for the formation of solid MX directly form elements According to Hess's law of heat summation U - the lattice enthalpy for the formation of solid MX ˆ†H 3- Ionisation energy for M(g) to M +(g) ∆H 4 - electron affinity for the conversion of X(g) to X -(g) 
ΔH 2- enthalpy change for the dissociation of ½ X 2 (g) to X(g) ˆ†H 1 - enthalpy change for the sublimation M(s) to M(g) For example consider the formation of a simple ionic solid such as an alkali metal halide MX, the following steps are considered. The equation is: Lattice enthalpy Heat of formation- Heat of sublimation- (1/2 Dissociation energy) - (sum of Ionization energies)- (sum of Electron Affinities) How do you calculate. The Born–Haber cycle applies Hess's law to calculate the lattice enthalpy. Lattice Energy is directly proportional to the Charge on ion and inversely proportional to radius of atom. The cycle is concerned with the formation of an ionic compound from the reaction of a metal with a halogen or other non-metallic element such as oxygen.īorn–Haber cycle is primarily used in calculating lattice energy, which cannot be measured directly. It was named after two German scientists Max Born and Fritz Haber who developed this cycle. 9.The Born–Haber cycle is an approach to analyse reaction energies.Calculate the lattice energy for CaF 2.Įnergy of formation for one mole of CaF 2 from its elements = -1228 kJ/molīack to Periodic Trends and Ionic Compounds The sublimation energy for Sr is 164 kJ/mol, E i1 = +549.5 kJ/mol, E i2 = +1064.2 kJ/mol, E ea for Cl (g) = -348.6 kJ/mol, energy for the formation of one mole of SrCl 2 from its elements = -803.7 kJ/mol, and the bond dissociation energy for Cl 2 (g) = +243 kJ/mol.Įxercise 5. Which has the highest lattice energy, Na 3P or Na 2S?Įxercise 4. Order the following ionic compounds from lowest to highest lattice energy. What is the lattice energy for MgF 2?Įxercise 2. Consider the following Born-Haber cycle for MgF 2. Worksheet: Ionic Compounds, Born-Haber Cycle, and Lattice EnergyĮxercise 1. A more extensive list can be found in other tables or in the Handbook of Physics and Chemistry. In the case of NaCl and MgCl 2, the MgCl 2 has the larger lattice energy because the magnesium cation is smaller, and the charge is a +2 rather than a +1 for the sodium ion.īelow is a table with the lattice energies of some ionic compounds. This is because the charges are the same, but the potassium ion is larger than the sodium ion. For example, the lattice energy of NaCl is larger than the lattice energy of KCl. Lattice energies are large when ions are closer together, the distance between the ions is small and when the charges are larger. 
Coulomb’s law is equal to a constant, k, multiplied by the product of the ion charges, z 1 and z 22, between the ions. Recall, lattice energy is positive meaning it is endothermic. The stronger the bond, the higher the lattice energy. Lattice energy, E lattice is dependent on the strength of the bond between the cation and anion in an ionic bond. The lattice energy is always positive, because it takes energy to separate the ions from the solid. The equation for the lattice energy is the reverse of the equation in Step 5 in the figure below, for the formation of the solid from its ions which releases 787 kJ/mol of energy.Ī Born-Haber cycle allows the calculation of the lattice energy for a solid ionic compound. The process absorbs energy, and is highly endothermic. The NaF distance in NaF, which has the same structure as KF, is 231 pm. The lattice energy of KF is 794 kJ/mol, and the interionic distance is 269 pm. Lattice energy, E lattice is the energy required to separate one mole of a solid ionic compound into its gaseous ions. kJ/mol Ionization energy for K 419 kJ/mol Electron affinity of Cl 349 kJ/mol Bond energy of Cl2 239 kJ/mol Energy of sublimation for K 90. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
